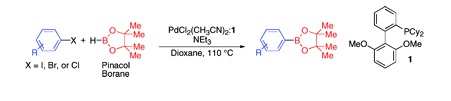
An Improved System for the Palladium-Catalyzed Borylation of Aryl Halides with Pinacol Borane
If you need a non-commercial boronic acid, you can follow two approaches: trapping of an organometallic (lithium or magnesium) with a boron source or palladium coupling with bis(pinacolato)diboron. The first method cannot always be used owing to the presence of sensitive groups and is usually long (generation of the organometallic; trapping with B(OMe)3, B(OiPr)3, or similar; and hydrolisis to boronic or conversion into a stable boronate). The second method, a Suzuki–Miyaura coupling, uses an expensive boron source, bis(pinacolato)diboron.
Buchwald (Cambridge, Mass, USA) has addressed this problem with the aim to find a cheaper boron source, cheaper starting materials, and use less catalyst. The final result is a method where any aryl halide (poor or rich, chloride, bromide, or iodide) can be used as the substrate. The catalytic system uses PdCl2(CH3CN)2, Sphos, and Et3N in dioxane at 110 °C. Depending on the halogen, the conversion can take up to 24 h. Yields are usually higher than 70%.
J. Org. Chem., 2008, 73 (14), pp. 5589–5591. See: 10.1021/jo800727s