A Fluorinated Ligand Enables Room-Temperature and Regioselective Pd-Catalyzed Fluorination of Aryl Triflates and Bromides
The development of a new phosphine ligand allows the room temperature fluorination of (hetero)aryl triflates and bromides.
The development of a new phosphine ligand allows the room temperature fluorination of (hetero)aryl triflates and bromides.
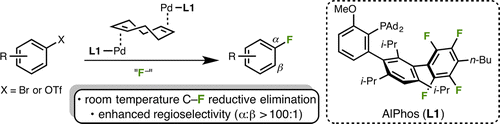
After a long, long time, I can finally squeeze some time for a new update on the blog. And I am updating with a new paper by Buchwald (MIT, MA, USA), dealing with the Palladium catalyzed fluorination of aromatics. Buchwald has already done a lof ot work in this field, but there is still (as usual) plenty of room for improvement. With the experimental and theoretical knowledge gathered they tweaked the ligands, and they have come up with a new biaryl monophosphine christened as AlPhos. This ligand is itself a fluorinated molecule, though this is just to confer the desired electronic properties to the ligand.
With the ligand at hand, they have run the usual set of experiments, coupling (hetero)aryl triflates with a fluorine source, CsF. The reaction takes days at r.t., but it works, giving yields above 80%. But you can increase the temperature a bit and run the reaction in 24 h while lowering the catalyst loading. An added benefit of the new ligand is that avoids the formation of regioisomers previously observed, allowing the use of the less reactive (hetero)aryl bromides which the previous reaction conditions were troublesome substrates.
In summary: if you can prepare the ligand and the preformed catalyst, here you have a new, good tool to fluorinate your compounds.
J. Am. Chem. Soc. 2015, Article ASAP.
See: 10.1021/jacs.5b09308