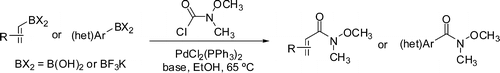
Palladium-Catalyzed Preparation of Weinreb Amides from Boronic Acids and N-Methyl-N-methoxycarbamoyl Chloride
A method for the preparation of Weinreb amides by palladium-catalyzed cross-coupling.
The Weinreb amides (N-methoxy-N-methylamides) are relatively new players in the organic synthesis arena. But in the brief time they have been present they have become increasingly important, because they have the remarkable ability to undergo a single substitution reaction in the presence of excess organometal reagents with minimal byproduct formation. In other words, adding ethylmagnesium bromide gives the ethyl ketone instead the alcohol. There are many protocols for the formation of these amides, ranging from the classical N,O-dimethylhydroxylamine addition to carboxylic acids up to some cross-coupling methods.
The work by Herr (AMRI USA) is another palladium-catalyzed cross-coupling method, but with an important addition: it makes full use of the last developments in the field to put together a very versatile protocol which takes advantage of the many boron building blocks available. In a typical protocol, one equivalent of a boronic acid is mixed with N-methoxy-N-methylcarbamoyl chloride (200 mol%), PdCl2(PPh3)2 (5 mol%) and K3PO4·H2O (200 mol%) as base in anhydrous ethanol at 65 °C for 2 hours. The reaction works also in other solvents, like toluene and THF, an important point when solubility issues arise. Yields are usually excellent.
J. Org. Chem., 2010, 75 (4), pp 1251–1258. See: 10.1021/jo902647h