Transition-Metal-Free O-, S-, and N-Arylation of Alcohols, Thiols, Amides, Amines, and Related Heterocycles
Introduction of nucleophiles using superbase.
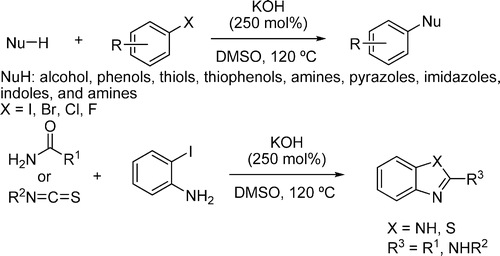
Looks like an Ullmann, but it is not. Looks like a SNAr, but it is not. Honestly, is one of the most surprising reactions I have seen for a long time, specially in view of the really simple reaction conditions. In a few words, Ramón et al. (Univ. of Alicante, Spain) have discovered that mixing an aryl halide (almost any aryl halide) with a nucleophile (choose one) and KOH (250 mol%) in DMSO at 120 °C leads to the substitution of the halide.
The reaction works with amines (aliphatic and aromatic), amides, phenols, alcohols, thiols and thiophenols. The mechanism must be really strange, because when pentafluoroiodobenzene is used as substrate, the substituted halide is the iodine!! The authors have discarded the contribution of metals to the mechanism analysing the presence of Copper and Palladium in the reaction system. Really amazing…
J. Org. Chem., 2011, 76 (2), pp 654–660. See: 10.1021/jo1022052