Regioselective Synthesis and Slow-Release Suzuki–Miyaura Cross-Coupling of MIDA Boronate-Functionalized Isoxazoles and Triazoles
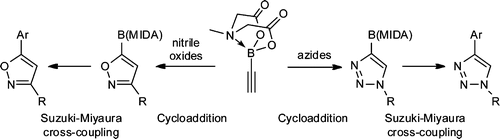
Preparation of boronic acid MIDA esters of isoxazoles and triazoles.
During the last CPhI I had the opportunity to meet with the people from Borochem, in France. They were announcing a partnership with Boron Molecular, an old supplier of us. They told me that their catalogs overlapped only a 10-20%, because they were using different technologies to produce the boronates/boronics. They have several thousand compounds in stock, but even with such number of available building blocks usually we have problems when we are preparing a library and we are looking for boronates/boronics of small heterocycles.
This paper by Grob et al. (Novartis, USA), describes a nice method to prepare different MIDA boronates of Isoxazoles and triazoles. To summarize, they take ethynyl boronic acid MIDA ester and perform a series of cycloadditions with oximes and azides to obtain the functionalizated heterocycles in good yields. They make use of the usual array of modern ligands, paladacycles and so, but the method is still interesting if you are working with those five membered rings.
J. Org. Chem., Article ASAP. See: 10.1021/jo201973t