Innate C-H trifluoromethylation of heterocycles
Introduction of the trifluoromethyl moiety into heterocycles.
I admit that I almost missed this paper. Honestly, the PNAS is not a journal I check every day, but this paper made some impact on other sites, so I became aware of it by a second hand source. And it is a good thing, because is one of those papers that many med chems will love. Just consider this: trifluoromethylation of heterocycles with a cheap, stable reagent. Man, that’s an instant hit !!!
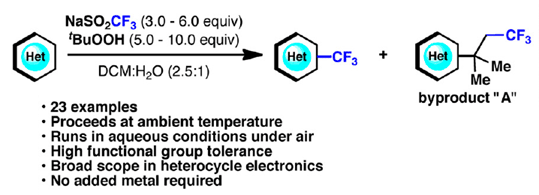
To summarize, using a very simple, cheap protocol developed by Baran et al. (The Scripps Research Institute, CA, USA), you can introduce the CF3 moiety into many heterocycles. More than 25 examples are presented, most of them pyridines and other pi-defficient heterocycles, with yields going from moderate to good. The reaction can be used on gram scale, though it has some drawbacks, the main one being that usually you obtain a byproduct derived from the reaction of the CF3 radical, the oxidant (tBuOOH) and the heterocycle. Other times there is no selectivity. But if you consider the advantages… no doubt about the usefulness of this new protocol. The paper is also nicely written, which at least for me is a bonus. See more work by Baran in this issue of our newsletter.
PNAS, 2011, 108 (35), pp 14411-14415. See: 10.1073/pnas.1109059108