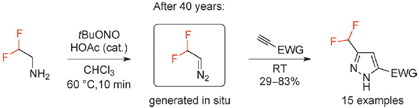
In Situ Generation of Difluoromethyl Diazomethane for [3+2] Cycloadditions with Alkynes
Safe preparation of an elusive reagent, difluoromethyl diazomethane, and its use for the preparation of pyrazoles.
I love very much this excerpt: “It is rather surprising that at a time when chemists can synthesize extremely complex compounds in more than 100 chemical steps, a very small reagent with only two carbon atoms and true potential for the synthesis of pharmaceuticals is still unknown.” As we have commented before in this blog, some people think that we can do everything in organic synthesis. But as the author notes, sometimes is incredible that some very simple products are impossible to prepare. Or maybe not.
So Pavel Mykhailiuk (Enamine, Ukraine) tackled the challenge without hesitation. After some experiments, he concluded that the trick to prepare the elusive product is to avoid water and basic medium. And that’s all. As usual, seeing the problem once it has been solved looks deceptively simple. The diazocompound is generated in solution, even in grams scale, and reacted wih a pool of alkynes to yield with excellent results a series of pyrazoles with a consistent regiochemistry. And though pyrazoles are intrinsically interestering, the potential of this reagent goes beyond this specific case. Wait and see.
Angew. Chem. Int. Ed. 2015, 54, pp 1-5.
See: 10.1002/anie.201501529